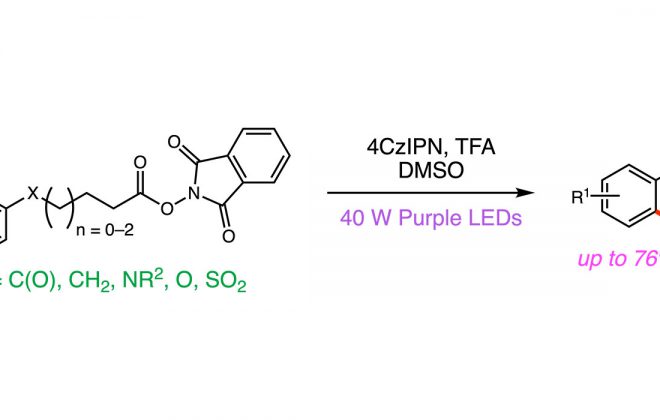
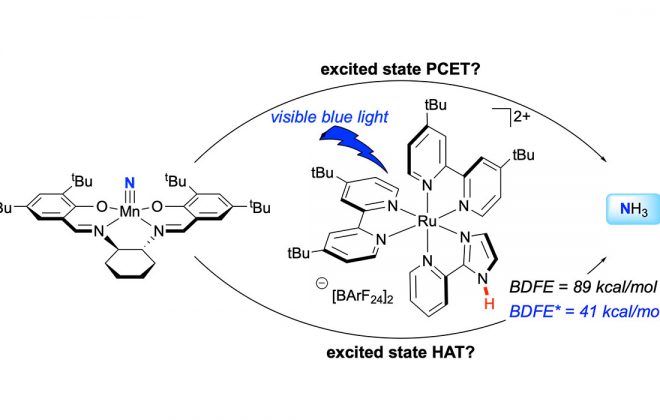
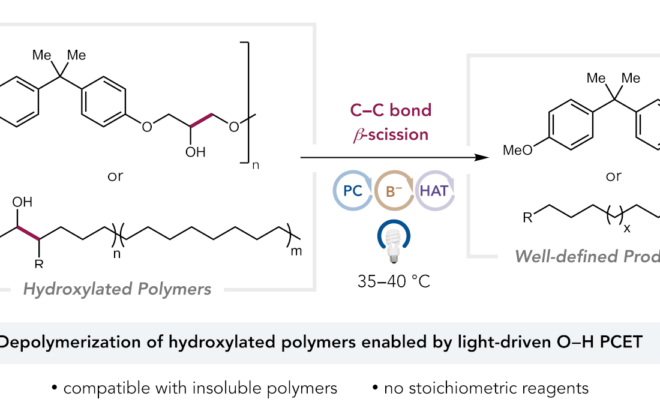
2. Enantioselective Photoredox Catalysis Enabled by Proton-Coupled Electron Transfer: Development of an Asymmetric Aza-Pinacol Cyclization
Lydia J. Rono, Hatice G. Yayla, David Y. Wang, Michael F. Armstrong, Robert R. Knowles:
https://pubs.acs.org/doi/abs/10.1021/ja4100595
ABSTRACT: The first highly enantioselective catalytic protocol for the reductive coupling of ketones and hydrazones is reported. These reactions proceed through neutral ketyl radical intermediates generated via a concerted proton-coupled electron transfer (PCET) event jointly mediated by a chiral phosphoric acid catalyst and the photoredox catalyst Ir(ppy)2(dtbpy)PF6. Remarkably, these neutral ketyl radicals appear to remain H-bonded to the chiral conjugate base of the Brønsted acid during the course of a subsequent C–C bond-forming step, furnishing syn 1,2-amino alcohol derivatives with excellent levels of diastereo- and enantioselectivity. This work provides the first demonstration of the feasibility and potential benefits of concerted PCET activation in asymmetric catalysis.