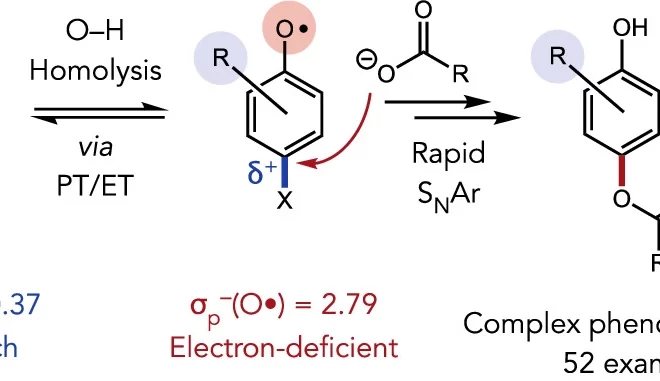
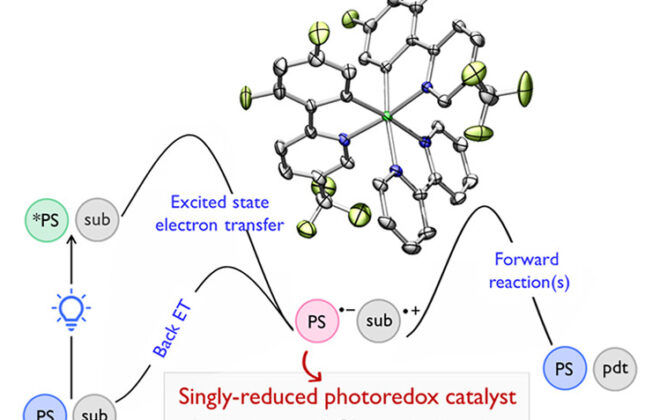
5. Bond-Weakening Catalysis: Conjugate Aminations Enabled by the Soft Homolysis of Strong N–H Bonds
Kyle T. Tarantino, David C. Miller, Ted A. Callon, Robert R. Knowles:
https://pubs.acs.org/doi/abs/10.1021/jacs.5b03428
ABSTRACT: The ability of redox-active metal centers to weaken the bonds in associated ligands is well precedented, but has rarely been utilized as a mechanism of substrate activation in catalysis. Here we describe a catalytic bond-weakening protocol for conjugate amination wherein the strong N–H bonds in N-aryl amides (N–H bond dissociation free energies ∼100 kcal/mol) are destabilized by ∼33 kcal/mol upon by coordination to a reducing titanocene complex, enabling their abstraction by the weak H-atom acceptor TEMPO through a proton-coupled electron transfer process. Significantly, this soft homolysis mechanism provides a method to generate closed-shell, metalated nucleophiles under neutral conditions in the absence of a Brønsted base.